Quantifying infectious disease threats is the first step toward controlling and eliminating them. Thousands of reports of possible infectious disease outbreaks are produced every month around the world, creating an urgent need to prioritize responses to those threats that are most pressing. Accurate measurement of the number of persons affected, their demographics, the transmissibility and severity of the infection, and the likely variation of these quantities over space and time is essential for evidence-based decisions about which responses to prioritize. Accurate evaluation of the impact of interventions such as vaccination and treatment is critical to ensure that decisions continue to reflect the best evidence.
Quantifying disease and intervention impact
Developing targeted strategies for malaria elimination
Malaria still kills nearly half a million people each year, mostly young children. Despite some success in reducing cases and deaths over the last 20 years, significant challenges remain for national malaria control programs in the control and elimination of this vector-borne parasite. Dr. Buckee’s group develops mathematical models to understand various aspects of malaria transmission, particularly with respect to targeting interventions for elimination strategies on a national level.
Dr. Buckee’s team is working with country-level control programs and researchers to develop methods to target interventions as malaria transmission is controlled and elimination becomes a priority. In particular, they have focused on novel approaches to vector control, developing models to understand the impact of new tools on population level epidemiology with the lab of Prof. Catteruccia (Dept of Immunology and Infectious Disease), and on the development of epidemiological models directly relevant for the malaria elimination agenda. Many of these approaches combine different data layers (e.g. see Malaria genetics section) to produce risk maps, which help programs make decisions about resource allocation. The group also works with National Malaria Control Programs in Bangladesh, Guyana, Mynamar and Thailand to develop analytical pipelines and risk mapping tools to aid their decision making for malaria control and elimination.
A major focus of the work is understanding how infections are imported between different regions of a country; reducing local transmission requires vector control, whereas catching imported infections requires surveillance and treatment. Targeting resources like bednets is therefore dependent on finding the foci of transmission that are exporting infections to the rest of the country. The new data sources from mobile phones, as well as parasite genetic data are used to model how this spatial spread occurs.
Key publications
Disrupting Mosquito Reproduction and Parasite Development for Malaria Control.PLoS Pathog. 2016.
Targeting Human Transmission Biology for Malaria Elimination. PLoS Pathog. 2015.
Wolbachia infections in natural Anopheles populations affect egg laying and negatively correlate with Plasmodium development. Nat Commun. 2016.
Measuring the association between artemisinin-based case management and malaria incidence in southern Vietnam, 1991-2010.Am J Trop Med Hyg. 2015.
A sticky situation: the unexpected stability of malaria elimination. Philos Trans R Soc Lond B Biol Sci. 2013
Modeling the human infectious reservoir for malaria control: does heterogeneity matter? Trends Parasitol. 2013
Insecticide resistance is threatening the most important interventions we have against malaria: insecticide treated bed nets and indoor residual sprays. The mosquito has evolved resistance to insecticides in almost every natural population measured, but the mechanisms underpinning the emergence of resistance and the dynamics of its emergence are poorly understood. Furthermore, with only crude assays for resistance in the field, even measuring its prevalence is challenging. In collaboration with entomology labs and field researchers, we are developing mathematical frameworks of mosquito population dynamics that allow us to track how resistance mutations can emerge under different scenarios, and to develop more nuanced assays for insecticide resistance. We also work with entomology labs who are developing new vector control tools, developing population models of mosquitoes transmitting malaria to understand how these new tools may impact the epidemiology of the disease.
Key publication
Disrupting Mosquito Reproduction and Parasite Development for Malaria Control. PLoS Pathog. 2016
Lastly, with health economists, we are also aiming to incorporate human behavior more accurately in our models of malaria transmission. In particular, how people perceive their risk of malaria depends on the clinical incidence of the disease, which is nonlinearly related to infection prevalence. Therefore, the utilization of bednets and other malaria interventions may not depend only on coverage of a particular intervention, but also on uptake by populations at risk. We are incorporating the relationship between perceived risk and intervention uptake into the predicted efficacy of control strategies for elimination.
Mathematical and statistical methods to measure the impact of infectious disease and control measures
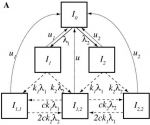 Infectious diseases pose special challenges for modeling and statistical analysis because transmission makes infection of one individual a risk for infections of others. Also, many infectious disease cases are never diagnosed, so methods are needed to infer who was infected and who was protected by particular interventions. Finally, the existence of multiple strains in pathogen populations complicates modeling inference about transmission dynamics.
Infectious diseases pose special challenges for modeling and statistical analysis because transmission makes infection of one individual a risk for infections of others. Also, many infectious disease cases are never diagnosed, so methods are needed to infer who was infected and who was protected by particular interventions. Finally, the existence of multiple strains in pathogen populations complicates modeling inference about transmission dynamics.
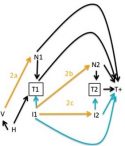
CCDD researchers have developed a variety of methods to enhance the analysis of infectious disease data, including negative controls for epidemiological studies, neutral models for multi-strain pathogens, and improved ways to estimate disease burden and identify core groups in transmission.
Key publications
No coexistence for free: neutral null models for multistrain pathogens. Epidemics. 2009
Negative controls: a tool for detecting confounding and bias in observational studies. Epidemiology. 2010
Observational studies and the difficult quest for causality: lessons from vaccine effectiveness and impact studies. Int J Epidemiol. 2016
Temporally Varying Relative Risks for Infectious Diseases: Implications for Infectious Disease Control. Epidemiology. 2017
Improving the estimation of influenza-related mortality over a seasonal baseline. Epidemiology. 2012
Clinical trial preparedness
Expediting vaccines in public health emergencies
The West African Ebola outbreak of 2014-5 underlined the need to devise faster, more efficient and ethical means of testing vaccines and treatments during public health emergencies.

A major thrust of current CCDD work is to develop new approaches and study the performance of existing trial designs for evaluating vaccines during outbreaks, especially of novel diseases. This work involves collaboration of mathematical modelers, biostatisticians, and ethicists as well as clinical trialists to find solutions that will give reliable results fast and in an ethical fashion.
Key publications
Improving vaccine trials in infectious disease emergencies. Science. 2017
Using simulation to aid trial design: Ring-vaccination trials. PLoS Negl Trop Dis. 2017
Competing Effects Of Indirect Protection And Clustering On The Power Of Cluster-Randomized Controlled Vaccine Trials. Am J Epidemiol. 2018
Modeling Vaccine Trials in Epidemics with Mild and Asymptomatic Infection. Preprint. 2018
Enhancing vaccine trials with new data sources [*cross listed in Harnessing Pathogen Evol.- Harnessing Evol. sub category]
CCDD researchers have been at the forefront of efforts to use pathogen sequence data to infer who infected whom, and separately to enhance vaccine trial designs (see above). We are now combining these streams to devise new ways to conduct vaccine trials while leveraging who-infected-whom inferences to measure vaccine effects in new ways and with greater precision.
Separately, we are designing ways to estimate vaccine effectiveness and correlates of protection simultaneously in clinical trials.
Simulation for trial design
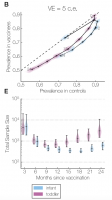 We develop computational methods to simulate complex clinical trials of infectious disease interventions.
We develop computational methods to simulate complex clinical trials of infectious disease interventions.
Studying the effects of measures to prevent and treat infectious diseases – whether behavioral interventions, vaccines, or antiinfective drugs – presents unique challenges because of the potentially multiple effects of the intervention (and sometimes the trial to study it) on transmission of the infection. Simulations can aid in the design, analysis and interpretation of clinical trials of vaccines and other countermeasures for infectious diseases. We also apply similar methods to study the properties of observational study designs, with the goal of improving their design and interpretation.
Key publications
Simulations for designing and interpreting intervention trials in infectious diseases. BMC Med. 2017
Impact of stochastically generated heterogeneity in hazard rates on individually randomized vaccine efficacy trials. Clin Trials. 2018
Quantifying biases in test-negative studies of vaccine effectiveness. Preprint. 2018
Displacement of sexual partnerships in trials of sexual behavior interventions: A model-based assessment of consequences. Epidemics. 2017
Host and pathogen heterogeneity
Heterogeneous red blood cell dynamics and malaria severity
The mechanisms that determine clinical outcomes of malaria are poorly understood, and range from asymptomatic or mild ‘flu-like symptoms to severe anemia, coma, and death. All symptoms of malaria occur during the blood stage of infection, when the parasite undergoes successive rounds of invasion of red blood cells, followed asexual growth, and cell lysis. The dynamics of blood-stage infection rely on host-parasite interactions that mediate invasion via specific pairs of parasite ligands and host receptors. Red blood cells themselves are also heterogeneous across hosts, and dynamic over the course of their 4-month lifespans with respect to available receptors for invasion. A fundamental basis for determining malaria virulence, therefore, is the extent to which red blood cells are available and receptive to invasion. We work with molecular geneticist Prof. Duraisingh (Dept of Immunology and Infectious Disease) to combine experiments and theoretical models of within-host dynamics of the malaria parasite to investigate how red blood cell heterogeneities and other host-parasite interactions impact the ability of the parasite to grow in vivo.
Key publications
Recent advances in the molecular epidemiology of clinical malaria. F1000Res. 2018
Resistance to Plasmodium falciparum in sickle cell trait erythrocytes is driven by oxygen-dependent growth inhibition. Proc Natl Acad Sci U S A. 2018
A Plasmodium falciparum histone deacetylase regulates antigenic variation and gametocyte conversion. Cell Host Microbe. 2014
Expansion of host cellular niche can drive adaptation of a zoonotic malaria parasite to humans. Nat Commun. 2013.
Heterogeneous susceptibility infection and protection by vaccination
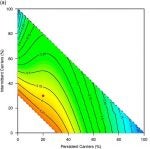 Individuals in any population vary in their susceptibility to infection by a given pathogen, and in their natural history of infection once infected. We quantify the impact of variable susceptibility to infection among individual hosts on the effectiveness of vaccination and other countermeasures. This work has application from hospital-acquired bacterial infections to viral infections of trout.
Individuals in any population vary in their susceptibility to infection by a given pathogen, and in their natural history of infection once infected. We quantify the impact of variable susceptibility to infection among individual hosts on the effectiveness of vaccination and other countermeasures. This work has application from hospital-acquired bacterial infections to viral infections of trout.
Key publications
Vaccine Effects on Heterogeneity in Susceptibility and Implications for Population Health Management. MBio. 2017
A missing dimension in measures of vaccination impacts. PLoS Pathog. 2014
Impact of Host Heterogeneity on the Efficacy of Interventions to Reduce Staphylococcus aureus Carriage. Infect Control Hosp Epidemiol. 2016
Across scales
The population structure and other characteristics of many pathogens vary in space and time, and for many reasons. For example novel drug resistant threats may emerge, or different strains of malaria parasite might be present in different locations. CCDD scientists work with collaborators around the world to define and track these changes, and to try to understand what has made them happen.
Key publications
Frequency-dependent selection in vaccine-associated pneumococcal population dynamics. Nat Ecol Evol. 2017
Genomic Epidemiology of Penicillin-Nonsusceptible Pneumococci with Nonvaccine Serotypes Causing Invasive Disease in the United States. J Clin Microbiol. 2017
Productive disruption: opportunities and challenges for innovation in infectious disease surveillance. BMJ Glob Health. 2018
Microevolution
All living things accumulate mutations as they copy their genetic material, by multiple mechanisms which can be used to study disease transmission (link to shared variants), and can give rise to new functions and adaptation in short time scales. In CCDD, we study both, trying to improve the use of genetic data to make inferences about transmission or import of an infection, and more fundamental research on the different ways pathogens have of accumulating variation, and its significance.
Key publications
Use of Genome Sequencing to Define Institutional Influenza Outbreaks, Toronto, Ontario, Canada, 2014-15. Emerg Infect Dis. 2018
Substitutions of short heterologous DNA segments of intragenomic or extragenomic origins produce clustered genomic polymorphisms. Proc Natl Acad Sci U S A. 2016
Comparative genomics of recent Shiga toxin-producing Escherichia coli O104:H4: short-term evolution of an emerging pathogen. MBio. 2013
Expository reviews
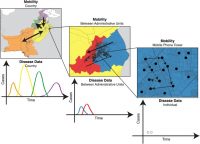
We strive to write clear, readable reviews of areas of infectious disease methods to assist researchers in designing and analyzing studies to maximize their value.
Key examples
Simulations for designing and interpreting intervention trials in infectious diseases. BMC Med. 2017
Choices in Vaccine Trial Design for Epidemics of Emerging Infections. Preprint. 2018
Potential Biases in Estimating Absolute and Relative Case-Fatality Risks during Outbreaks. PLoS Negl Trop Dis. 2015
Improving vaccine trials in infectious disease emergencies. Science. 2017
Cholera modeling: challenges to quantitative analysis and predicting the impact of interventions. Epidemiology. 2012
Temporally Varying Relative Risks for Infectious Diseases: Implications for Infectious Disease Control. Epidemiology. 2017
Antibiotics in agriculture and the risk to human health: how worried should we be? Evol Appl. 2015
Origin and proliferation of multiple-drug resistance in bacterial pathogens. Microbiol Mol Biol Rev. 2015
Seasonal Population Movements and the Surveillance and Control of Infectious Diseases. Trends Parasitol. 2017
Connecting Mobility to Infectious Diseases: The Promise and Limits of Mobile Phone Data. J Infect Dis. 2016
Not So Simple After All: Bacteria, Their Population Genetics, and Recombination. Cold Spring Harb Perspect Biol. 2016
Interim results of an ecological experiment – Conjugate vaccination against the pneumococcus and serotype replacement. Hum Vaccin Immunother. 2016